EXPAREL is the widely used and very popular post-operative pain treatment for large joint arthroplasty. But not for spine. Have two Chicago doctors created the alternative to EXPAREL for spine surgery…and possibly any other surgical intervention as well?
EXPAREL
Tom Zdeblick, Chair of the University of Wisconsin Department of Orthopedics, Dan Riew, then Professor of Orthopedic Surgery at Washington University School of Medicine, Sig Berven, Associate Professor at the University of California, Alex Vaccaro, Professor of Orthopaedics at Thomas Jefferson University and Stephen Hochschuler, past-president of the Spine Arthroplasty Society volunteered to review submissions for the Best New Spine Technology award for 2013.
Buried in about 500 pages of product and technology information was EXPAREL’s application.
EXPAREL, for those who don’t know, is a single administration liposomal formulation of bupivacaine. Its purpose is to reduce post-operative pain. It works by extending bupivacaine’s pain relieving ability for 24 – 48 hours. And it does this by encapsulating bupivacaine in a form of phospholipid (fat globules) known as multivesicular liposome or DepoFoam.
Multivesicular liposomes have been used for decades to affect a controlled release of nutrients or drugs in many applications.
In its submission to the 2013 Spine Award judges, EXPAREL’s manufacturer wrote that EXPAREL had been used to manage post-operative pain in such spine procedures as lumbar, cervical and interbody fusions; microdiscectomies; and laminectomies.
The judges were impressed and named EXPAREL one of the ten winners for 2013.
The Rise of Post-Operative Pain Management in Orthopedics
In 2013, the year EXPAREL won the award as a Best Spine Technology, its supplier, Pacira Pharmaceuticals, Inc., sold about $86 million of EXPAREL.
Two years later—2015—that number had tripled to $240 million—59% of which came from orthopedics—and most of that was for large joint arthroplasty.
Wall Street thinks that Pacira Pharmaceuticals is worth $1.8 billion.
Pacira has really found a big win with its re-packaged bupivicaine. But, more importantly, improving post op pain management fits into the broader trends of shorter hospital stays, more rapid rehabilitation and, icing on the cake, much less opioid use.
Happy patient. Happy surgeon. Happy hospital administrator.
Oh…and with EXPAREL patients rated their overall experience higher.
ReLeaf Anesthetic Delivery Catheter
Neel Anand, Director of Orthopaedic Spine Surgery at Cedars-Sinai, Scott Blumenthal, assistant professor of orthopedic surgery at the University of Texas Southwestern, Alan Hilibrand, Professor of Orthopaedic Surgery and Neurosurgery at Jefferson Medical College, Daniel Riew, then Professor of Orthopedic Surgery at Washington University School of Medicine and Rick Sasso, Associate Professor and Chief of Spine Surgery at the Indiana University volunteered to review submissions for the Best New Spine Technology award for 2014.
Buried in about 500 pages of product and technology information was ReLeaf’s application.
ReLeaf is a directional analgesic infusion catheter and wound drainage system. ReLeaf continuously infuses the surgical site with anesthesia for up to (and potentially beyond) 72 hours post-surgery.
ReLeaf delivers anesthesia in a controlled release fashion to the surgical site during the post-operative recovery period. Like EXPAREL. But not like EXPAREL.
ReLeaf is a controlled release catheter while EXPAREL is a pharmacological solution.
Both are FDA cleared.
Both provide effective pain relief during those first couple, three days after surgery.
Both replace the need for opioids in that post-operative recovery period.
Both are highly cost effective (reduced opioid use, shorter hospital stays and as a percent of the total cost of surgery).
But one you can turn off and one you can’t.
Background
In 2002, Dr. Frank Phillips then at The University of Chicago Hospitals decided to test the effects of continuous local anesthetic agent infusion at the ICBG harvest site in a prospective randomized, double blind study. When he moved to Rush University Medical Center, Dr. Phillips as Minimally Invasive Spine Surgery Section Director and Professor, Department of Orthopedic Surgery and Kern Singh, then Assistant professor, Department of Orthopedic Surgery compiled the data.
In other clinical procedures—most popularly child birth—continuous local anesthetic infusions are very effective at reducing pain. Why not for iliac crest bone graft?
So Drs. Phillips and Singh reported on 37 spine fusion surgery patients. Average age 60. Randomized to either saline or bupivacaine (0.5% Marcaine).
Both groups received treatment for 48 hours (96 mL [2 mL/hr × 48 hours] of either normal saline [control group, n = 22] or 0.5% Marcaine [treatment group, n = 15] delivered via a continuous infusion catheter placed at the ICBG harvest site).
It worked.
After 48 hours of continuous infusion of bupivacaine (0.5% Marcaine) at the surgical site patients reported significantly reduced pain scores and a 50% drop in postoperative parenteral narcotic usage. No complications were attributed to the infusion-catheter system.
Here’s the Crazy Part
“I thought he was crazy, ” remembers Dr. Phillips. Dr. Singh suggested that the team do a four-year follow up.
Sure enough, the team found 19 patients from the original study group who were willing to return and be tested again.
To be clear, bupivacaine is out of the patient’s body in about 24 hours. A four-year follow up is roughly 35, 000 hours after administration. This looked like an empty exercise.
But it wasn’t. In fact, it showed a continuous and strong difference between the treated and control group.
Here’s the direct quote from the follow up study—which was published in Spine (Spine 2007;32 (25):2790-2796).
Results: Nine of 11 patients (82%) in the treatment group and 10 of 14 patients (71%) in the control group were available at final follow-up (1 death occurred in the control group unrelated to the study). The treatment group had a statistically significant decrease in the graft site pain VAS score (1.4 vs. 4.8) and increased satisfaction with the procedure at a minimum of 4 years post procedure (P < 0.05). Additionally, no patient in the treatment group developed chronic iliac crest dysesthesias (0 of 9) versus 7 of 10 patients (70%) in the control group (P < 0.05).
Ok. Four years later. The treated group STILL had less graft site pain than the control group. And none of the treated patients had chronic iliac crest dysesthesias versus 70% of the control patients.
Crazy, right?
In an attempt to explain this clearly unanticipated result Phillips and Singh wrote the following:
“The extended beneficial effect of reduced chronic donor site pain observed in this investigation was not anticipated. This may be suggestive of a preemptive analgesic effect in which a reduction in spinal cord hyperexcitability is attained by a prompt reduction in noxious afferent input.
Direct application of local anesthetic to wounds can provide analgesia through several mechanisms. There is growing evidence that local anesthetics can inhibit chemical response to injury, a noxious process that may sensitize nociceptive receptors and contribute to chronic pain and hyperalgesia. Studies have observed that local anesthetics reduce the release of inflammatory mediators from neutrophils, reduce neutrophil adhesion to the endothelium, reduction formation of free oxygen radicals, and decrease edema formation potentially reducing the incidence of chronic pain. One of the inevitable side effects of ICBG harvesting is pain at the donor site with chronic pain being reported in up to 39% of cases. Various studies have suggested that the use of local anesthetic at the ICBG site reduces postoperative pain and facilitates a quicker return to daily activities.”
The Invention
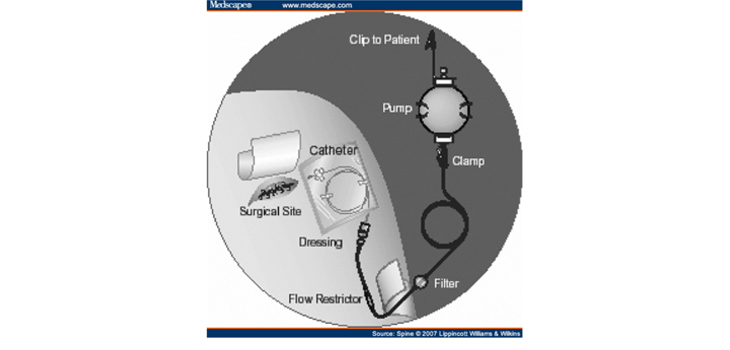
In the original study, the two doctors used an off-the-shelf I-Flow catheter to deliver the bupivacaine infusion. But off the shelf catheters were not designed specifically for this purpose.
So Phillips and Singh together designed a dual function directional analgesic infusion catheter with a wicking sponge, which ALSO allowed the wound site to drain. They then partnered with inveterate engineer/inventor, Wade Fallin, and the three of them developed ReLeaf.
Fallin, Phillips and Singh created a catheter with a semi-permeable barrier technology. In spine surgery in particular, this helped to minimize local anesthetic contact with exposed neural elements.
The inventors named their invention ReLeaf and formed a company named Vital 5 to continue the development and eventual commercialization of the dual function catheter.
The clinical applications for the Vital 5 ReLeaf include any invasive surgical procedure, including spine, orthopedic, cardiothoracic, plastic, general, obstetrics and gynecological procedures.
Ok, Where Can We Get this?
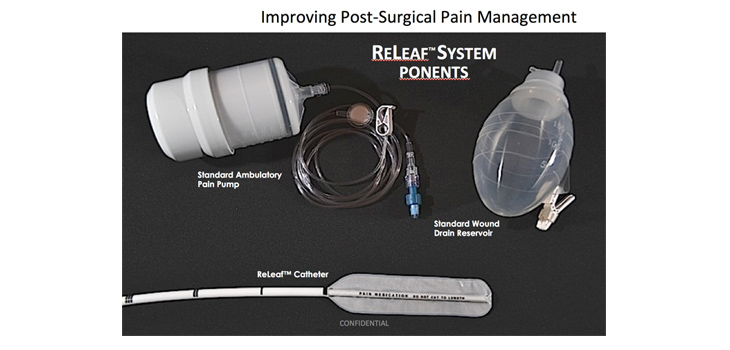
It’s not on the market, yet. But with FDA clearance in hand it is only a matter of time. Already the new catheter has been used in dozens of spinal and large joint replacement applications.
According to the two doctors, this approach will, in the words of Dr. Singh, “bend the cost curve”. Not only is the cost of the catheter itself low, but it demonstrated in the small study to cut opioid use by 50% and, as we’ve seen with the EXPAREL experience, should also be able to cut time in the hospital and improve rehab.
Most interesting, of course, is the long term effect of applying a continuous infusion of bupivacaine to the wound site immediately after surgery. The four-year data is extremely interesting and should be tested further at multiple sites and many more patients. Something is going on here.
Last, but not least, is that a catheter infusion approach has one obvious advantage over the pharmacological approach—it can be turned off. In big deformity cases, open lumbar fusion cases—really anywhere that the surgeon has concern over nerve damage—the infusion catheter can be turned on and off at any time to allow for neurologic assessment, whereas with long acting anesthetic applied at the time of surgery there is no off switch.
For more information: email Tony Recupero at: tony@catalystperformanceadvisors.com.