In what may be good news for back pain sufferers resulting from its clinical trial of ReActiv8, Mainstay Medical International plc reports lasting improvement in pain, disability and quality of life for people with chronic low back pain (CLBP). Between 63% and 72% of the participants in the study reported clinically important improvement in their back pain.
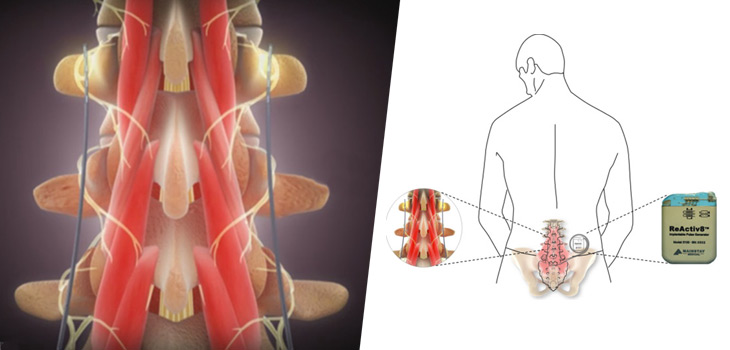
ReActiv8 is spinal cord stimulation, the company explains—a new implantable neurostimulation system—that “targets different clinical conditions and delivers electrical stimulation to interfere with the perception of pain, without addressing the root cause.” The market, says Mainstay’s management is about 100, 000 patients annually.
Business Wire reports that participants in the trial had a mean age of 43.9 years and a long history (3.8 years) of low back pain. All had tried physical therapy and 70% were taking opioids. The results of the trial are based on data from the first 47 subjects implanted in the ReActiv8-A Trial. Forty-six of them reached the 90 day follow up and to date 33 have reached the 180 day follow up.
Dr. Marc Russo, Director of the Hunter Pain Clinic in Newcastle, Australia, and an investigator in the trial said: “It was pleasing to see ReActiv8 have such an impact on people’s quality of life after so many other conventional treatments have been unsuccessful for such a long time.”
Peter Crosby, CEO of Mainstay, added: “The results from the ReActiv8-A Trial show improvements which are better than any other therapy for this group of people as reported in the literature. We are excited that our unique approach to treating this type of chronic low back pain offers the potential to change the lives of millions of people worldwide who have no effective treatment alternative.”
Mainstay officials say there were no unanticipated adverse events (AEs) and no serious AEs related to the device, therapy or procedure. The company plans to apply for a CE Mark for ReActiv8. Subjects continue to be enrolled in the ReActiv8-A Trial to gather additional data on performance and safety which the company plans to incorporate into the Post Market Clinical Follow Up.


Am interested to know if we can get this device privately
What is the cost ?