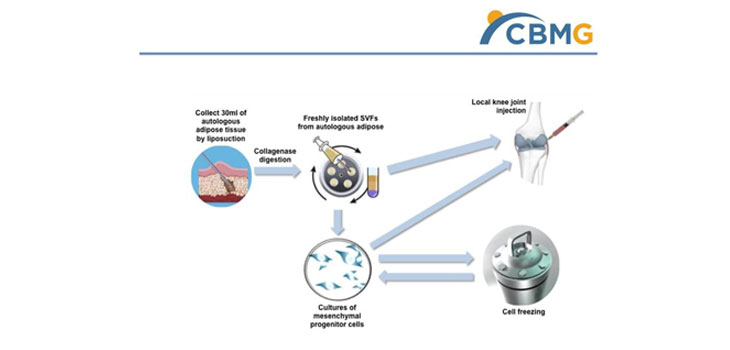
An updated analysis of a Phase 1/11a clinical trial of adipose-derived mesenchymal precursor cell therapy for knee osteoarthritis (KOA) showed no serious adverse effects and some encouraging results. Participants experienced significantly reduced knee pain, improved knee mobility and prolonged walking distance.
Cellular Biomedicine Group, Inc. presented the results of its trial at the New York Stem Cell Summit in February. Shanghai’s Renji Hospital, a tertiary hospital affiliated with Shanghai Jiao Tong University School of Medicine, conducted the trial. Researchers observed a reduction of bone lesions in some patients while MRI examination revealed an increase in cartilage thickness as early as three months after the therapy.
Data on three patients who completed the six-month follow-up confirmed the three-month findings. A full analysis of the six-month follow-up for all patients in the trial is still ongoing.
Wei Cao, PhD, BM, CEO of Cellular Biomedicine Group, Inc. said, “We are very excited with the preliminary analysis of the trial. As far as we know this may be the first evidence that adipose derived stem cells can regenerate human cartilage in three months. Our preliminary conclusion from the three-month follow-up of the Phase I/IIa clinical trial is that ReJoin cell therapy for KOA patients is safe.”
Cao noted that there are approximately 57 million people in China suffering from knee osteoarthritis. As drug-based methods of management are ineffective, life quality of KOA patients is compromised, and many patients with this disease will degenerate to the point of requiring invasive artificial joint replacement surgery.