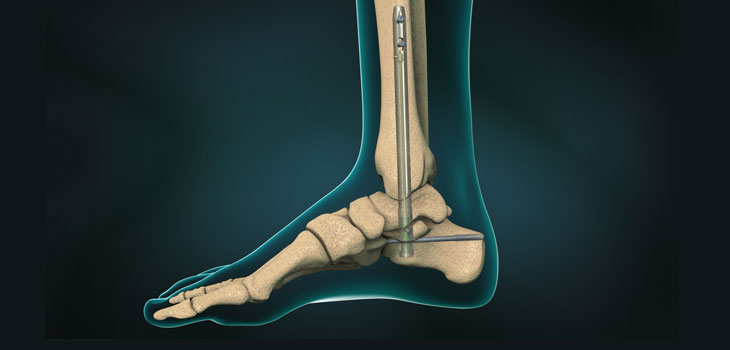
MedShape, Inc. is reporting on the first wave of clinical outcomes in patients who have received the DynaNail TTC Fusion System. To date during its targeted soft launch period, DynaNail has been successfully implanted in over 100 tibiotalocalcaneal (TTC) fusion procedures. Because the internal nickel titanium (NiTiNOL) element maintains the target fusion bones in close apposition and under sustained compression, fusions are being observed with the DynaNail TTC Fusion System in high-risk patients, specifically those who require bone allografts.
However, a large number of high-risk patients have experienced fusions with DynaNail, as confirmed by CT scan, including many with bulk allografts. Dr. Thomas San Giovanni of the UHZ Sports Medicine Institute in Coral Gables, Florida has implanted DynaNail in five patients, using a femoral head allograft for three, with successful fusion in each.
“I believe we may be entering a new era within orthopedics where the unique properties of certain materials such as NiTiNOL will be used to our advantage to assist in the healing of bone—complementing both the mechanical and biologic nature of bone healing, ” said Dr. San Giovanni in the January 7, 2014 news release. “The DynaNail is the first product of its kind and certainly is on the brink of this technology. I’ve had very good success with the DynaNail in some of the most difficult clinical scenarios where combined arthrodesis of the ankle and subtalar joint was needed. It has become my preferred fixation method when using a nail for TTC fusions. I have been very impressed by its performance and foresee the technology and unique properties of this nail lending itself to many future applications, even for other conditions.”
DynaNail is the only TTC fusion device to harness the superelastic properties of NiTiNOL. Its internal NiTiNOL element allows for compression to be maintained across the joint post-operatively by automatically adapting to loading changes due to settling or resorption. In addition, the ultra-low axial stiffness of the NiTiNOL element automatically dynamizes the joint, and mitigates stress-shielding that is universal to all other intramedullary nails on the market.
Kurt Jacobus, CEO of MedShape told OTW, “We are excited to have some of the best surgeons in the world using our unique technology to benefit patients with debilitating foot and ankle conditions.”
Asked about the ‘hard’ release, Jacobus said, “We are planning a multi center clinical study at Duke and an increase of sets in the marketplace to benefit a broader range of patients. A year from now we want to be part way through the Duke clinical study aimed at understanding high fusion rates and resulting benefits of the Dynanail to patients.”