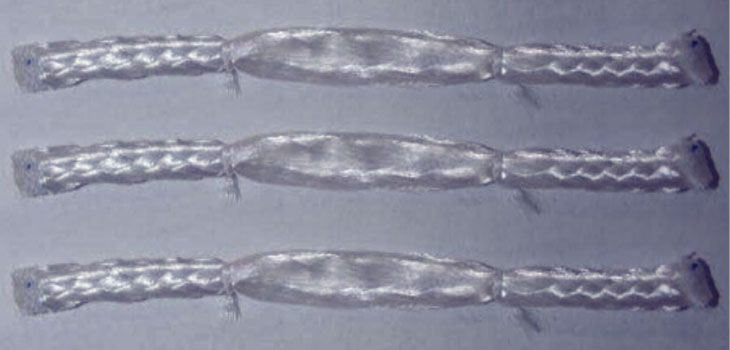
Soft Tissue Regeneration, Inc. (STR) has announced that it was issued a U.S. patent related to the L-C Ligament, a device for soft tissue regeneration of the anterior cruciate ligament (ACL) of the knee. The device was invented by Cato T. Laurencin, M.D., Ph.D., and is a bioresorbable scaffold that is designed to regenerate knee ligament tissue after ACL reconstruction surgery.
The patented technology uses a clinically proven degradable polymer called poly(L) lactide acid (PLLA) to address known risks and morbidity associated with allograft and autograft tissues. The technology requires no harvesting of the patient’s tendon, which eliminates the risks associated with the harvest site. The L-C Ligament encourages the regeneration of the patient’s own ligament tissue. To date, results from large-scale animal testing at one year and longer have demonstrated that the L-C Ligament can successfully regenerate a native ligament intra-articularly.
The L-C Ligament is an investigational device and is not available for use or sale in the European Union or the United States. It is only available as part of the clinical trial.
STR President and CEO Joseph Reilly told OTW, “We have completed all animal studies needed to justify our recent ‘first in man’ surgery on June 18 as part of a 15 patient safety study being conducted in Europe. However, we currently have eleven (11) sheep that were implanted with the L-C Ligament as part of a long term animal study. Three (3) of these sheep were implanted 3 years and 3 months ago and eight (8) of the sheep were implanted 2 years and 2 months ago. All of these sheep are doing fine and have ambulated normally since the day of their surgery. We are developing a program to bring some of these sheep out for as long as 5 years. It should also be noted that with almost 90 sheep that were implanted with the L-C Ligament, not one ever failed in vivo.”
Asked about having a patent, but not being able to sell the device in the U.S., Reilly stated, “In July of 2013, we submitted a Pre-Submission filing to the FDA to begin the process of obtaining IDE [investigational device exemption] approval to begin a clinical trial in the USA. After receiving 12 month data from our EU safety study in late 2014, we expect to file that information with the FDA and to receive IDE approval in 2015. Then we will begin a clinical trial in the USA that would allow Soft Tissue Regeneration to eventually obtain pre market approval (PMA) to market the L-C Ligament in the USA.”