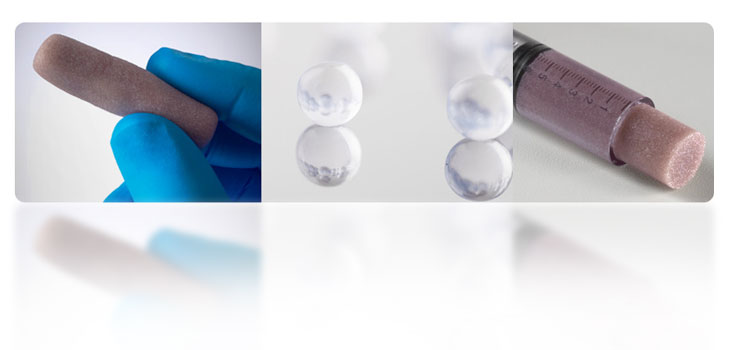
Bone putty made of spherical glass particles has received FDA clearance, according to its manufacturer, Synergy Biomedical, LLC, and is launching its first bone graft product called BioSphere Putty. Company officials claim that BioSphere, which uses a specialized form of 45S5 bioactive glass with a unique sphere shape, provides an optimized bone graft material that outperforms other bioactive products.
Among claims for the product is that the spherical shape of the BioSphere particles provides uniform and repeatable resorption and ion release. The 3-D packing of the spheres in a bone defect creates an optimal porosity that allows bone formation throughout the implant. Biosphere Putty is composed of bioactive glass spheres that are combined with a moldable phospholipid carrier. Company officials say that the putty has the highest bioactive glass composition on the market.
“The use of spherical particles has allowed Synergy to identify an optimal form of bioactive glass that improves bone formation, ” stated Mark Borden, Ph.D., CEO and founder of Synergy Biomedical. Established in 2011, Synergy Biomedical is a privately held medical device company focused on bringing biomaterial based products to the orthopedic and spinal markets.