The U.S. Food and Drug Administration (FDA) has granted 510(k) clearance to a trochanteric nailing system.
The nailing system, per the 510(k) summary document, is intended for “use in fracture fixation in the femur in adults with osteopenia or osteoporosis.” According to the 510(k) summary document, the nailing system is indicated for use in the following:
- Intertrochanteric and subtrochanteric fractures
- Segmental fractures
- Comminuted fractures
- Pathological fractures
- Fractures with bone loss
- Pseudoarthrosis, non-union, mal-union, and delayed union
- Surgically created defects such as osteotomies
In order to qualify for the shorter and earlier 510(k) a device must be substantially equivalent to a predicate device. This new femur fixation nailing system is substantially equivalent to earlier cleared intramedullary hip nail systems for fracture fixation in the femur. The predicate device, in fact, was also from the same manufacturer, Dublin, Ireland-based X-Bolt Orthopedics.
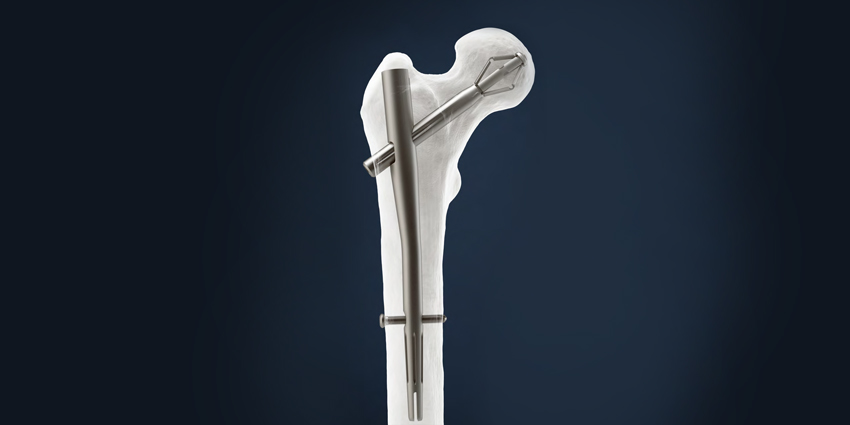
The newly cleared device is brand named the Pro-X1™ Trochanteric Nailing System.
The nailing system is a single use device intended for long-term implantation into the femur. It consists of a metal expanding hip bolt; a metal intramedullary nail that is available in various lengths; a set-screw that prevents the bolt from rotating but allows for “dynamic movement”; and interlocking screws that are used in conjunction with the nail to “achieve distal fixation.”
The components and instrumentation are what make the Pro-X1 Trochanteric Nail unique.
According to the company’s press release, the system “features a titanium nail construct with a reversibly expanding bolt instead of a typical lag screw.” The expanding bolt allows for “stronger anchorage and rotational stability of the femoral head.” Its instrumentation includes a patented “curved jig with flexi-drive coupling, to streamline the operative technique.”
The company plans to have the system’s commercial launch before the October 2023 Orthopaedic Trauma Association (OTA) meeting. According to the company’s press release, the Pro-X1 will be the company’s “flagship launch in the United States.”
X-Bolt Orthopedics Founder and CEO Brian Thornes commented, “We are thrilled to receive FDA clearance for the Pro-X1 Trochanteric Nail.”
Thornes continued, “Pro-X1 builds off of the foundational expanding bolt design, clinically proven with a 0.8% cut-out rate in the largest ever hip fracture randomized controlled trial. We are proud to offer this innovative device to orthopedic trauma patients and are confident that it will make a significant impact in the lives of those in need.”