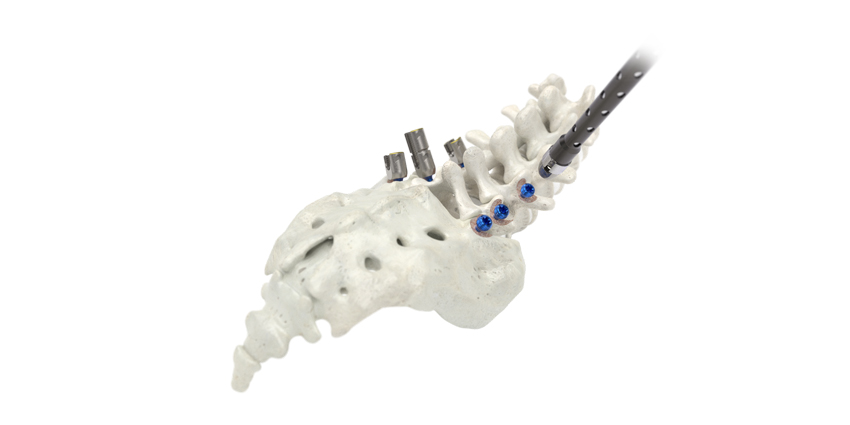
SeaSpine Holdings Corporation’s Mariner Cap System, “intended to provide temporary stabilization as a bone anchor during the development of solid bony fusion and aid in the repair of bone fractures,” has been 510(k) cleared by the FDA.
According to FDA clearance documents, the system is to be used in conjunction with the company’s pedicle screw system, “whenever ‘wiring’ may help secure the attachment of other implants.”
The Cap system, with its three components—the Mariner Cap SP Connector, the Mariner Cap SP Locking Base, and the Locking Insert—combines SeaSpine’s pedicle screw system with France-based Implanet’s JAZZ Cap technology. The companies initiated this partnership 18 months ago.
Indications
“The indications for use include the following applications:
- Spinal trauma surgery, used in sublaminar or facet wiring techniques;
- Spinal reconstructive surgery, incorporated into constructs for the purpose of correction of spinal deformities such as adolescent idiopathic scoliosis, adult scoliosis, kyphosis and spondylolisthesis;
- Spinal degenerative surgery, as an adjunct to spinal fusions. “
Mariner Cap System
The Cap is designed to “mate” with the Mariner screws and is added to the proximal end of the Mariner Screw. This, according to the documents, “…provides for an alternate method to attach the JAZZ Band and JAZZ Passer Band to skeletal structures as compared to the current method using a JAZZ connector.”
The Cap system provides, “a more efficient, lower profile method of adding the braid attachment to the fixation construct. The locking mechanism used for the braid component in the Mariner Cap is identical to that already cleared in the JAZZ CAP.”
SeaSpine, Implanet Partnership
SeaSpine President and CEO, Keith Valentine, said in an October 19, 2020 press release, “The Mariner Cap System further strengthens the company’s range of products dedicated to treating spinal disorders, and represents a proprietary alternative that we can offer our distributors, surgeons and their patients across the U.S. Combining our two technologies provides a unique solution for securing the stability of pedicle screw constructs. We look forward to continuing our innovative partnership with Implanet.”
SeaSpine obtained the distribution rights for the JAZZ range under its own brand name in the U.S.
It took the FDA about three months to clear the system.