Your patient presents with pain and swelling in the knee. She says that her joint is stiff, and her range of motion has declined.
Arthritis?
She also tells you that the skin over her knee feels warm and tender. Her swollen knee is painful, and she tells you she is experiencing functional limitations in her daily life.
A possible diagnosis could be tenosynovial giant cell tumor (TGCT), which is typically a non-malignant tumor. While the exact incidence of TGCT is not known, it is estimated that the worldwide incidence of TGCT is 43 patients per million person-years.1
But when it occurs, it can cause the type of debilitating symptoms mentioned above and can be locally aggressive, reducing mobility in the affected joint or limb.2,3,4
Two Types of TGCT
TGCT is classified into two types: diffuse TGCT or localized TGCT.
For patients with localized TGCT, the more common form of TGCT which typically affects smaller joints, surgery can usually remove the tumor, though up to 15% of patients experience recurrence after complete resection.5,6
For patients with diffuse type TGCT, which is less common but more aggressive, joints tend to harbor larger tumors and in larger joints, which makes the surgical option, resecting the tumor, more difficult.4,7,8 Additionally, diffuse TGCT has a higher rate of recurrence of up to 55%.4,7,8
For healthcare providers and patients, it’s crucial to understand the differences between the types of TGCT, how each can affect patients’ lives and how to determine treatment plans.
Not all Patients With Symptomatic TGCT Are Amenable to Improvement With Surgery
TGCT can be life-altering, causing chronic pain and adversely impacting function and everyday activities.7,8,9 But that doesn’t mean that one treatment fits all patients.
“Healthcare providers treating TGCT have limited treatment options outside of surgical resection,” said Kamalesh Sankhala, M.D., Complex, Advanced, Rare Cancers and Sarcoma Medical Oncology Clinical Research and Experimental Therapeutics, Cedars Sinai Medical Center. “It is critical that we continue to explore treatment options for patients with TGCT for whom surgery may not be an option or may require multiple surgeries.”
A Non-Surgical Treatment Option for TGCT
The standard of care for treating TGCT is surgery to remove the tumor.4 However, certain patients with symptomatic TCGT may not be amenable to improvement with surgery. For these patients, TURALIO® (pexidartinib) may be considered. TURALIO is the first FDA-approved oral therapy for adult patients with symptomatic TGCT associated with severe morbidity or functional limitations and not amenable to improvement with surgery.10
TURALIO was approved by the FDA in 2019 and is believed to work by inhibiting CSF-1R, a key mechanism behind TGCT tumor formation and growth.10 As of December 2024, there have been over 750 patients treated with TURALIO since the launch.
“By inhibiting CSF-1R, TURALIO has been shown to help reduce tumor size,” said Dr. Sankhala. “For patients with symptomatic TGCT whose severe limitations can’t be improved with the current standard of care, TURALIO may offer a path forward to potentially reducing tumor size and improving range of motion.”
TURALIO can cause serious and potentially fatal liver injury, including vanishing bile duct syndrome. Patients must have liver tests prior to initiating TURALIO and be monitored at specified intervals during treatment. Treatment may need to be withheld, the dose reduced, or permanently discontinued if hepatotoxicity occurs. Monitoring and prompt cessation of TURALIO may not eliminate the risk of serious and potentially fatal liver injury. TURALIO is available only through a restricted program called the TURALIO Risk Evaluation and Mitigation Strategy (REMS) Program. TURALIO may cause fetal harm when administered to a pregnant woman. See additional Important Safety Information, including Boxed Warning below.
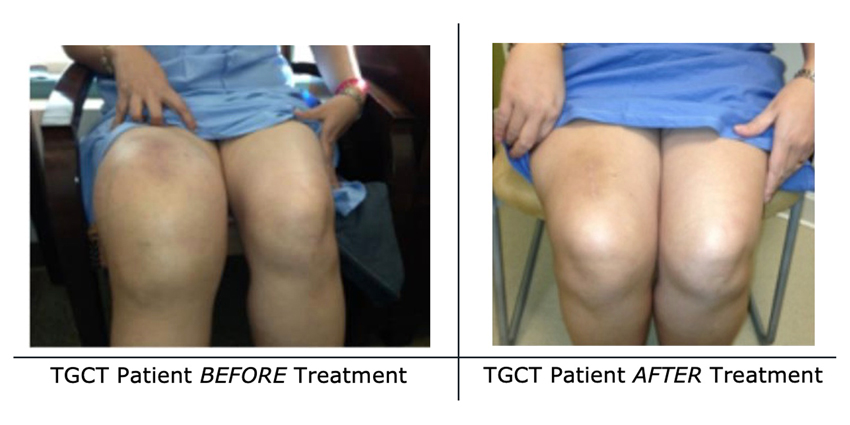
Before and After Photos
Here is an example of a patient’s experience and response to treatment with TURALIO. Each patient’s results will vary. The need to consider future surgery for TGCT, including amputation, may be warranted.
Before treatment (pictured left), the patient was unable to straighten her knee and experienced debilitating symptoms.
The photos on the right show the response the patient’s tumor had to TURALIO.

*Each patient’s results will vary. The need to consider future surgery for TGCT, including amputation, may be warranted.
The current recommended dose of TURALIO is 250 mg twice daily (taken as two 125 mg capsules) with a low-fat meal of approximately 11 to 14 grams of total fat until disease progression or unacceptable toxicity. Patients should consult with their healthcare providers to answer any questions they may have about TURALIO or the dosing regimen.
Individualizing Treatment Planning for TGCT Patients
Every patient is unique, and TGCT presents across a broad spectrum of cases. Developing a tailored, patient-centered treatment plan is essential to ensuring the best possible care.
While some TGCT patients may benefit from surgery, others may not be amenable to improvement with surgery. For these patients, TURALIO may be a potential treatment option. When discussing treatment plans with TGCT patients, it is important to provide a comprehensive overview of the risks and benefits of surgical resection as well as other available therapies.
By considering all available treatment options, healthcare providers can better support TGCT patients in finding the most effective path to managing their condition.
INDICATION
TURALIO® (pexidartinib) is indicated for the treatment of adult patients with symptomatic tenosynovial giant cell tumor (TGCT) associated with severe morbidity or functional limitations and not amenable to improvement with surgery.
IMPORTANT SAFETY INFORMATIOCONTRAINDICATIONS: None
WARNING: HEPATOTOXICITY

CONTRADICTIONS: None
WARNINGS AND PRECAUTIONS
- Hepatotoxicity, including liver failure and life-threatening vanishing bile duct syndrome (VBDS), ductopenia, and symptomatic cholestasis (including severe pruritus) can occur in patients treated with TURALIO and can occur despite monitoring and prompt drug cessation.
- The mechanism of cholestatic hepatotoxicity is unknown and its occurrence cannot be predicted. It is unknown whether liver injury can also occur in the absence of increased transaminases.
- Of the first 609 patients who received TURALIO under the REMS program, 32 (5.3%) developed a liver injury event of concern, defined as any serious liver-related outcome or any liver abnormality that triggers drug discontinuation per the US Prescribing Information. These 32 patients developed liver toxicity within 71 days of the first dose of TURALIO; ten required hospitalization and two developed VBDS. Sixteen of the 32 patients had not fully recovered at the time of the analysis, including 6 patients followed for at least 6 months after discontinuation.
- Among 768 patients who received TURALIO in clinical trials, there were two irreversible cases of cholestatic liver injury. One patient with advanced cancer and ongoing liver toxicity died and one patient with a confirmed case of VBDS required a liver transplant.
- In ENLIVEN, 3 of 61 (5%) patients who received TURALIO developed signs of serious liver injury, defined as alanine aminotransferase (ALT) or aspartate aminotransferase (AST) ≥3 × upper limit of normal (ULN) with total bilirubin ≥2 × ULN. In these patients, peak ALT ranged from 6 to 9 × ULN, peak total bilirubin ranged from 2.5 to 15 × ULN, and alkaline phosphatase (ALP) was ≥2 × ULN. ALT, AST, and total bilirubin improved to <2 × ULN in these three patients 1 to 7 months after discontinuing TURALIO.
- Avoid TURALIO in patients with preexisting increased serum transaminases, total bilirubin, or direct bilirubin (>ULN); or active liver or biliary tract disease, including increased ALP.
- Monitor liver tests, including AST, ALT, total bilirubin, direct bilirubin, ALP, and gamma-glutamyl transferase (GGT), prior to initiation of TURALIO, weekly for the first 8 weeks, every 2 weeks for the next month and every 3 months thereafter.
- Withhold and dose reduce, or permanently discontinue TURALIO based on the severity of the hepatotoxicity. Refer patients to a hepatologist if liver tests do not return to normal. Rechallenge with a reduced dose of TURALIO may result in a recurrence of increased serum transaminases, bilirubin, ALP or other signs of liver injury. Monitor liver tests weekly for the first month after rechallenge.
TURALIO REMS
- Requirements include: 1) prescribers must be certified by enrolling and completing training, 2) patients must complete and sign an enrollment form for inclusion in a patient registry, and 3) pharmacies must be certified and must dispense only to patients who are authorized (enrolled in the REMS patient registry).
- Further information is available at TURALIOREMS.com or 1-833-887-2546.
Embryo-Fetal Toxicity
- TURALIO may cause fetal harm when administered to a pregnant woman. Advise patients of reproductive potential of the potential risk to a fetus. Verify pregnancy status in females of reproductive potential prior to the initiation of TURALIO.
- Advise females of reproductive potential to use an effective nonhormonal method of contraception. TURALIO can render hormonal contraceptives ineffective during treatment with TURALIO and for 1 month after the final dose.
- Advise males with female partners of reproductive potential to use effective contraception during treatment with TURALIO and for 1 week after the final dose.
Potential Risks Associated with a High-Fat Meal
- Taking TURALIO with a high-fat meal increases pexidartinib concentrations, which may increase the incidence and severity of adverse reactions, including hepatotoxicity.
- Instruct patients to take TURALIO with a low-fat meal (approximately 11 to 14 grams of total fat) and to avoid taking TURALIO with a high-fat meal (approximately 55 to 65 grams of total fat).
ADVERSE REACTIONS
- The most common adverse reactions (>20%) were increased lactate dehydrogenase (92%), increased AST (88%), hair color changes (67%), fatigue (64%), increased ALT (64%), decreased neutrophils (44%), increased cholesterol (44%), increased ALP (39%), decreased lymphocytes (38%), eye edema (30%), decreased hemoglobin (30%), rash (28%), dysgeusia (26%), and decreased phosphate (25%).
DRUG INTERACTIONS
- Hepatotoxic products: Avoid coadministration in patients with increased serum transaminases, total bilirubin, or direct bilirubin (>ULN) or active liver or biliary tract disease.
- Moderate or strong CYP3A inhibitors and UGT inhibitors: Concomitant use may increase pexidartinib concentrations. Reduce TURALIO dosage if concomitant use cannot be avoided.
- Strong CYP3A inducers: Avoid concomitant use due to decreased pexidartinib concentrations.
- Acid-reducing agents: Avoid concomitant use of proton pump inhibitors due to decreased pexidartinib concentrations. Use histamine-2 receptor antagonists or antacids if needed.
- CYP3A substrates: Avoid concomitant use where minimal concentration changes may lead to serious therapeutic failure (e.g., hormonal contraceptives) due to decreased concentrations of CYP3A substrates.
USE IN SPECIFIC POPULATIONS
- Lactation: Advise not to breastfeed and for at least 1 week after the final dose.
- Renal impairment: Reduce the dosage for patients with mild to severe renal impairment.
- Hepatic impairment: Reduce the dosage for patients with moderate hepatic impairment. TURALIO has not been studied in patients with severe hepatic impairment.
To report SUSPECTED ADVERSE REACTIONS, contact Daiichi Sankyo, Inc. at 1-877-437-7763 or FDA at 1-800-FDA-1088 or fda.gov/medwatch.
Please see full Prescribing Information, including Boxed WARNING, and Medication Guide.
References
- National Organization for Rare Disorders. Tenosynovial Giant Cell Tumor. Accessed October 2024.
- de Saint Aubain, et al. 2013;100-103.
- Rao AS, et al. J Bone Joint Surg AM. 1984;66(1):76-94.
- Ravi V, et al. Curr Opin Oncol. 2011;23:361-366.
- Mastboom MJL, et al. Acta Orthopaedica. 2017;88(6):688-694.
- Ehrenstein V, et al. J Rheumatol. 2017; 44(10):1476-1483.
- Verspoor FGM, et al. Future Oncol. 2013;10:1515-1531.
- Verspoor FGM, et al. Rheumatol. 2014;53(11):2063-2070.
- Brahmi M, et al. Curr Treat Options Oncol. 2016;17(2):10.
- TURALIO [package insert]. Basking Ridge, NJ: Daiichi Sankyo, Inc; 2025.

© 2024 Daiichi Sankyo, Inc. PP-US-TU-1246 10/24