On November 4, 2020, NuVasive, Inc. announced its Thoracolumbar Interbody System had been FDA 510(k) cleared to include the treatment of multi-level sagittal deformities of the thoracolumbar spine. The clearance expands indications from one- to two-level spine surgery, to multiple levels, for the treatment of sagittal malalignment.
Sagittal Deformities
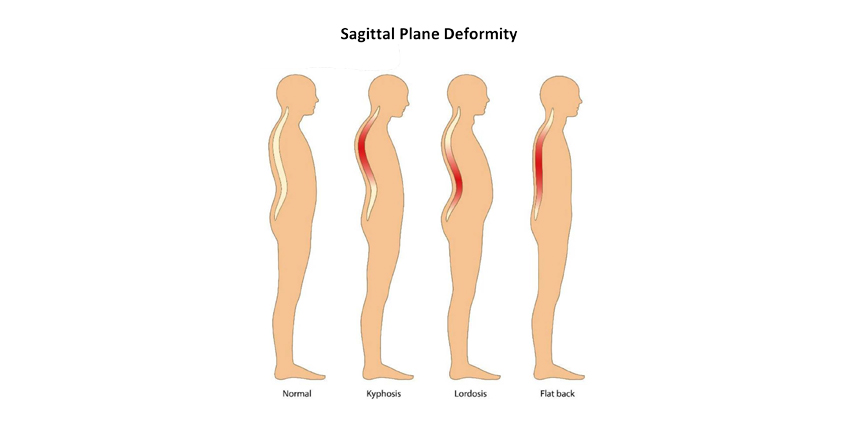
Sagittal deformities are one of the more common sources of pain and disability for patients and will affect the ability of a patient to stand upright. Aging is the most common cause of sagittal deformity, but more specifically, loss of disc height plus weakened back muscles contributes to stooped posture—a manifestation of sagittal deformity. When indicated, intervention by a surgeon usually requires removing a portion of the spine and implanting spinal fixation screws and rods in order to correct the sagittal plane deformity and restore normal lumbar lordosis. And, of course, relieve pain and restore function.
One-of-a-Kind Clearance
Company Executive VP Massimo Calafiore said, “NuVasive’s thoracolumbar interbodies are now the only implants on the market to be clinically validated in and indicated for the treatment of sagittal deformities.”
NuVasive’s Ever Growing Family of Interbody Systems
The impressive family of thoracolumbar interbody systems which are part of this FDA clearance are:….
- CoRoent Thoracolumbar System,
- CoRoent XL Interfixated System,
- Brigade Standalone System and Brigade Hyperlordotic System,
- Brigade Lateral System,
- BASE Interfixated Titanium System,
- Coalesce Thoracolumbar Interbody Fusion System,
- Cohere Thoracolumbar Interbody System,
- Modulus XLIF Interbody System,
- Modulus TLIF Interbody System, and,
- 3DP Interfixated ALIF System
NuVasive offers this extensive library of systems in a variety of dimensions which allow surgeons to adapt to the specific patient need and, according to the NuVasive’s marketing department, “can significantly improve alignment in the sagittal plane.”
But that is not all. NuVasive also provides the following enabling technologies to ensure that the surgeon meets all clinical goals, to wit:
- “Integrated Global Alignment (iGA) platform, comprised of surgical planning tools—NuvaLine and NuvaMa O.R.—designed to enhance clinical and economic outcomes by increasing the predictability of achieving global alignment in spinal procedures through pre-, intra-, and postoperative measurement of alignment; and,
- Bendini, a proprietary, digital spinal rod bending system used to create patient-specific rods to effectively restore sagittal alignment.”
All systems, according to FDA documents, “are intervertebral body fusion devices, interfixated and non-interfixated. Implants are manufactured of either PEEK-Optima LT-1(Polyether-ether-ketone) or Titanium alloy. PEEK implants include radiographic markers made of Titanium (Ti), or Tantalum (Ta). Interfixated implants include Titanium alloy screws. CoRoent Thoracolumbar Interfixated implants include canted coil locking mechanism of Nickel-Cobalt-Chromium-Molybdenum alloy.”
Indications
Each of the 10 of the interbody systems have their own indications.
“This recent clearance for expanded indications provides broader scientific justification for not only sagittal deformity correction with these implants, but also their benefit in less invasive spine surgery,” said Adam Kanter, M.D., associate professor of neurological surgery and director of the Minimally Invasive Spine and fellowship programs at the University of Pittsburgh School of Medicine, in Pittsburgh, Pennsylvania.