The FDA has 510(k) cleared IlluminOss Medical, Inc. to add the treatment of fractures of the fibula to the previously cleared treatment of traumatic, fragility, pathological, and impending pathological fractures of the humerus, radius, ulna, clavicle, pelvis, metacarpals, metatarsals, and phalanges.
Photodynamic Bone Stabilization System
The Photodynamic Bone Stabilization System, according to the company, is the world’s first patient conforming intramedullary implant and has been in clinical use in Europe since 2010, with over 4,000 procedures to date.
The device utilizes a light-curable liquid monomer, contained within an expandable balloon, to create a patient-conforming, rigid implant within the bone canal.
In an August 18, 2020 press release, Robert Rabiner, the company’s chief technology officer, said fractures of the fibula, one of the long bones of the lower leg, “are among the most common and can be particularly challenging to manage in elderly patients. Additionally, current treatment approaches such as plating have several disadvantages, including larger incisions and the potential for meaningful patient discomfort.
He added that the company’s technology offers a minimally invasive approach “that spares soft tissues, which may lower the risk of wound complications, and can potentially drive better functional outcomes, including faster return to weight bearing and activities of daily living.”
The IlluminOss technology is covered by over 110 issued patents in the U.S. and internationally, with additional patent applications pending.
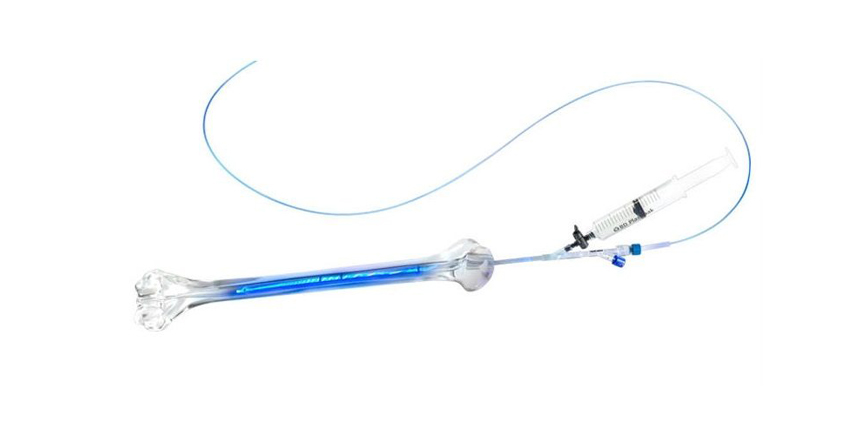
System Mechanics
According to FDA clearance documents, “the system uses a catheter to deploy an inflatable, noncompliant, thin wall PET [polyethylene terephthalate] balloon into the medullary canal of the bone across the fracture site. The balloon is infused using a syringe with a photodynamic (light cured) monomer that causes the balloon to slowly expand and fill the intramedullary canal of the fractured bone.”
Activation of the light system allows for visible spectrum light “to be delivered through a radially emitting light fiber that is temporarily positioned into a central lumen of the catheter that runs the length of the balloon. With this design, the liquid monomer within the balloon is exposed to light along the entire length of the balloon during the curing process.”
Mike Mogul, the company’s chairman, said returning elderly fracture patients quickly to activities of daily living remains a significant challenge or opportunity in orthopedics. “The fibula clearance, in addition to the recently approved pelvic and clavicle indications, is yet another step toward IlluminOss’ goal of providing minimally invasive fracture care in the majority of fractures faced by those elderly patients.”
HealthpointCapital Acquisition
On May 1, 2020, HealthpointCapital, announced the acquisition of a majority stake in the privately held company. Mike Mogul, a leader at HealthpointCapital, is former Group President, Orthopaedics at Stryker Corporation, and former CEO of DJO Global, a former portfolio company of Blackstone.